 
The following are three compounds whose pKa values we'll be examining:Īcids with a lower pKa value have a higher pKa value. An acid that has a lower number is considered stronger. 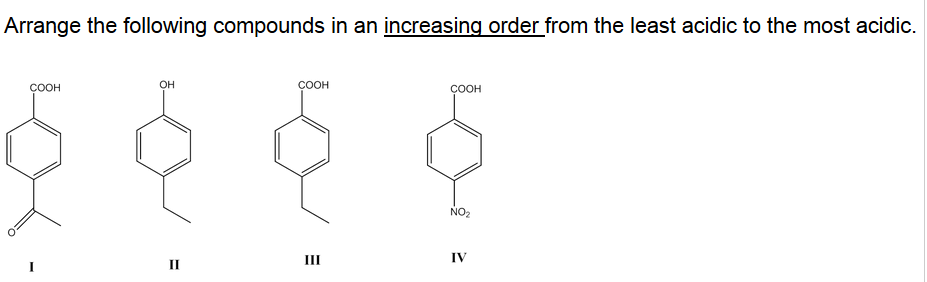
Changes in the properties or reactions of a molecule due to variations in its substituents are called substituent effects.ĭepending on their strength, weak acids are ranked on the pKa scale. The resonance effect causes the phenyl or vinyl groups on carboxylic acids to increase the acidity even though the inductive effect causes the acidity to decrease.Įffects of substituents on acidity Substituent effectsIn the context of discrete atoms or molecules, a substituent is known as an atom or molecule fragment. However, electron-donating groups destabilize the conjugated bases those carboxylic acids form, resulting in reduced acidity.ĬF3COOH > CCl3COOH > CHCl2COOH > NO2CH2COOH > NC-CH2COOH By adding the electron-withdrawing group to carboxylic acids, the conjugate base becomes more stable and, as a result, more acidic. Negative charges can be effectively delocalized by electron-withdrawing groups through resonance and inductive effects. Moreover, the acidity of carboxylic acids depends on the substituents attached to the carboxyl group, such as alkyl or aryl. The carboxyl group makes the group more acidic when electron withdrawal occurs, while electron donation makes the group less acidic. Metals and alkalis are capable of forming carboxylate ions only by resonating with carboxylic acids. Because acidic carboxylic acids are acidic, whereas alkaline phenols are alkaline. The stability of the phenoxide ion is thus greater. Two equivalent resonance structures of the carboxylate ion transfer the negative charge to two further electronegative oxygen atoms as a carboxylate ion.Ĭonversely, the phenoxide ion has a smaller area where the electrons are delocalized and the carbon atoms are less electronegative. Carboxylic acids have a higher acidity than alcohols and phenols. Organic compounds with carboxylic acids are stronger than their mineral counterparts, but that's not the case in the reverse. 
In a resonance effect, the negative charge of the carboxylate ion is effectively delocalized, stabilizing it. The carboxylate ion and the hydronium ion are formed during the dissociation of carboxylic acids in water. Carbonates are acidic because of the -COOH group of hydrogen ions. As a rule, a carboxylic acid donates its electron to a hydrogen atom. When carboxylic acids reacted with weak bases, such as carbonates and bicarbonates, carbon dioxide was formed, which was a stronger acidity than that of simple phenols. Similarly, acidic carboxylic acids are affected by these reactions. Hydrogen and metals combined in an alkaline solution produce carboxylate ions. The Acidity of carboxylic acidCarbaryl acid is the name given to organic compounds that contain carboxylic acid groups. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
